Clinical
Atropine - wonder or weak treatment?
Sponsored by
In this article:
First published: November 30, 2018
Updated: August 16, 2021
Atropine has been the apparent hero of myopia management since the 2006 publication of the ATOM (atropine for the treatment of childhood myopia) study, which showed an 80% refractive and 100% axial length myopia controlling effect with 1% atropine compared to placebo.1 The significant side effects of mydriasis and cycloplegia, though, made it less than tolerable as a first line treatment for progressive myopia.
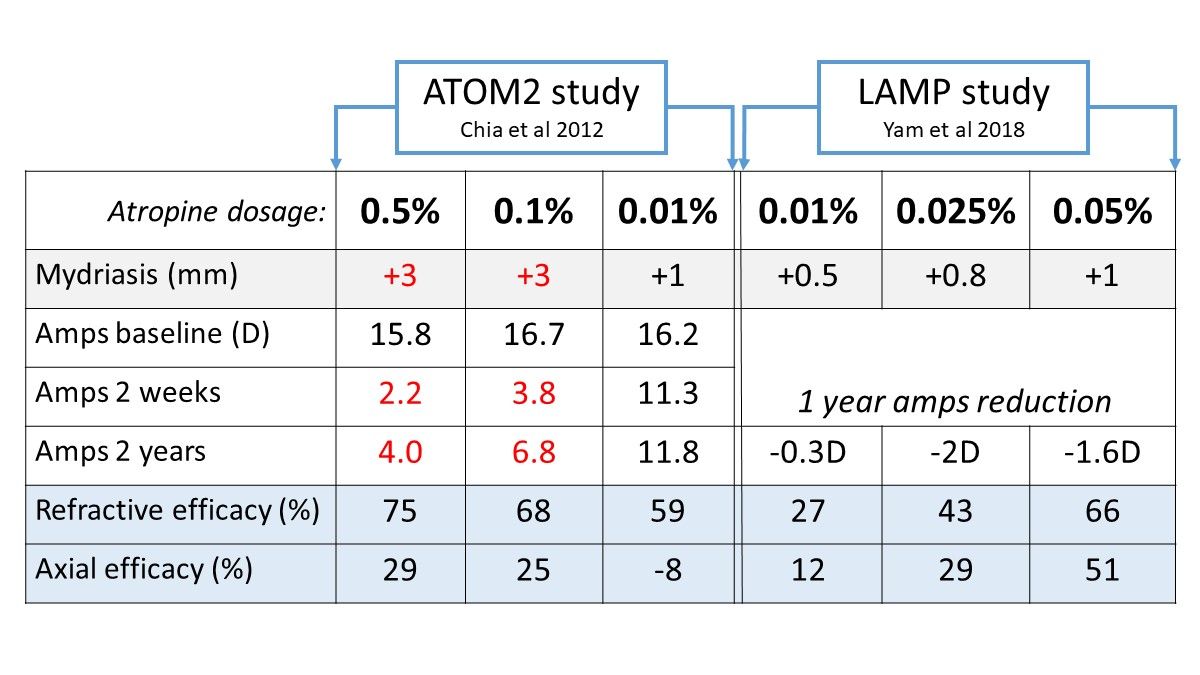
More recently, low dose (0.01%) atropine has become the new hero, on the basis of an evolving story in the literature. Firstly, the ATOM2 study in 2012 investigated 0.5%, 0.1% and 0.01% atropine for efficacy, and found that in both refractive and axial length control, the results appeared to be more impressive with greater concentrations.2 This study did not include a control group - as the story goes, the 0.01% atropine was meant to be the placebo. The plot twist was the ATOM2 five year study, published in 2016, which took the participants of the ATOM2 study and from year 2 to year 3, discontinued all treatment. Greater rebound effects were noted with the higher concentrations. Then, at the start of year 4, all remaining participants were recommenced on 0.01% atropine. The final picture at the end of year 5 showed that the children who had commenced on 0.01% atropine for two years, discontinued for a year, then commenced 0.01% again for another two years, had the lowest overall refractive progression.3 This low dose of atropine is much more clinically palatable, appearing to have a negligible side effect profile compared to higher concentrations.
When the crown slipped on 0.01% atropine
In early 2018, Mark Bullimore and David Berntsen published a letter to the Editor of JAMA Ophthalmology4 evaluating the data on low dose (0.01%) atropine, pointing out the enormous mismatch between its efficacy for refractive versus axial length control. In secondary analysis - comparison to the historical ATOM1, 2006 study control group - the ATOM2 data showed a 59% refractive control effect but a -8% axial length control effect for 0.01% atropine. If axial length is a key risk factor for risk of myopia pathology and vision impairment5 then this must bring into question the clinical usefulness of low dose atropine, which is clinically popular, especially in ophthalmology-driven myopia management.
A study with a concurrent control group is the better method for assessing comparative efficacy. In mid-2018, the Low-Concentration Atropine for Myopia Progression (LAMP) was published online, with one year data again showing efficacy correlated with concentration.6 More studies are underway around the world, and in other ethnicities, as all of this cited data has been collected on children in Asian countries.
Which atropine concentration is best right now?
Comparative efficacy and side effects of a variety of atropine concentrations are shown in the table above. As shown, 0.1% and 0.5% dilated the pupil by around 3mm and dramatically impaired near vision in the ATOM2 study - this makes these concentration an unpalatable option for school aged children who need comfortable near vision to learn. By comparison, 0.025% and 0.05% showed a small pupil dilation and minimal loss of accommodative amplitude in the LAMP study. Once again, 0.01% atropine looks pretty unimpressive as a first line therapy.
There's still a lot we need to learn about atropine, including any more subtle effects on binocular vision beyond amps of accommodation, ideal dosage schedules, treatment tapering and safe discontinuation. Given that current research indicates that 0.05% atropine has similar efficacy to optical interventions - namely, myopia controlling spectacles, dual-focus soft contact lenses and orthokeratology - in my mind it makes sense to consider one of these optical treatments as a first-line monotherapy where they are available to you and suitable to your patient. This is because optical treatments can both correct and control the myopia. Where these treatments aren't available to you or suitable to your patient, you can step through the options as outlined in our Clinical Decision Trees.
For a great summary of the ATOM1 and ATOM2 data in a clinician-accessible form, check out the June 2018 article written by Professor John Phillips of University of Auckland for the NZ Optics journal.
The plot thickens - new formulations and combination treatments
Not all atropine formulations are created equal - there are a handful of studies currently underway which are investigating novel preparation, low concentration atropine formulations designed to be more stable and consistent than compounded formulations. The first interim reports are likely due in late 2021, which you can read more about in The latest and greatest research on atropine. These new studies might change the message on 0.01% - just as different designs of myopia controlling soft contact lenses have different efficacy, the same may be proven of different formulations of low-concentration atropine.
The other interesting twist in the atropine story is combination treatments. A handful of studies of 1-2 years duration are indicating a likely role of 0.01% atropine combined with orthokeratology to increase myopia control efficacy. The picture is still being revealed on the ideal targets for this treatment, and for how long the combination treatment should last. Read more in Combination atropine treatments: when more is more.
Read the rest of our six-part series on atropine
Check out these clinical cases
You can also listen to our three podcasts on atropine with world-leading researchers
- Atropine, engaging with science and responsible practice with Professor Karla Zadnik from Ohio State University, USA.
- More on atropine 0.01% treatment for myopia management with Professor Mark Bullimore from the University of Houston, Texas USA.
- Atropine 0.01% for myopia management with Professor James Loughman from Technological University Dublin, and the Centre for Eye Research Ireland.
Meet the Authors:
About Kate Gifford
Dr Kate Gifford is an internationally renowned clinician-scientist optometrist and peer educator, and a Visiting Research Fellow at Queensland University of Technology, Brisbane, Australia. She holds a PhD in contact lens optics in myopia, four professional fellowships, over 100 peer reviewed and professional publications, and has presented almost 300 conference lectures around the world. Kate is the Chair of the Clinical Management Guidelines Committee of the International Myopia Institute. In 2016 Kate co-founded Myopia Profile with Dr Paul Gifford; the world-leading educational platform on childhood myopia management. After 13 years of clinical practice ownership, Kate now works full time on Myopia Profile.
This content is brought to you thanks to an educational grant from
References
- Chua W-H, Balakrishnan V, Chan Y-H et al. Atropine for the treatment of childhood myopia. Ophthalmol. 2006;113:2285-91. (link)
- Chia A, Chua W-H, Cheung Y-B et al. Atropine for the treatment of childhood myopia: safety and efficacy of 0.5%, 0.1%, and 0.01% doses (Atropine for the Treatment of Myopia 2). Ophthalmol 2012;119:347-54. (link)
- Chia A, Lu Q-S, Tan D. Five-Year Clinical Trial on Atropine for the Treatment of Myopia 2: Myopia Control with Atropine 0.01% Eyedrops. Ophthalmol 2016; 123;391-9. (link)
- Bullimore M, Berntsen D. Low-dose atropine for myopia control: considering all the data. JAMA Ophthalmol 2018;136:303. (link)
- Tideman JW, Snabel MC, Tedja MS et al. Association of Axial Length With Risk of Uncorrectable Visual Impairment for Europeans With Myopia. JAMA Ophthalmol 2016;134:1355-63. (link)
- Yam JC, Jiang Y, Tang SM et al. Low-Concentration Atropine for Myopia Progression (LAMP) Study: a randomized, double-blinded, placebo-controlled trial of 0.05%, 0.025%, and 0.01% atropine eye drops in myopia control. Ophthalmol 2019;126:113-24. (link)
Enormous thanks to our visionary sponsors
Myopia Profile’s growth into a world leading platform has been made possible through the support of our visionary sponsors, who share our mission to improve children’s vision care worldwide. Click on their logos to learn about how these companies are innovating and developing resources with us to support you in managing your patients with myopia.











